Turning a Self-Report Questionnaire into a Wearable Device
Well-defined and reliable tools are needed for objective diagnosis and to measure clinically relevant outcomes of treatment for myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS). The 2015 Institute of Medicine (IOM) report  “Beyond ME/CFS: Redefining an Illness” defines these symptoms for us namely, a substantial reduction or impairment in the ability to engage in pre-illness levels of occupational, educational, social, or personal activities, that persists for more than six months and is accompanied by fatigue, post-exertional malaise (PEM), unrefreshing sleep along with cognitive impairment or orthostatic intolerance.
“Beyond ME/CFS: Redefining an Illness” defines these symptoms for us namely, a substantial reduction or impairment in the ability to engage in pre-illness levels of occupational, educational, social, or personal activities, that persists for more than six months and is accompanied by fatigue, post-exertional malaise (PEM), unrefreshing sleep along with cognitive impairment or orthostatic intolerance.
Our goal at BHC is to take these criteria one-by-one and develop ways to measure them. Our medical experience over the past 17 years with ME/CFS patients has shown that asking patients to report the amount of time they spend in an upright position (defined as having lower legs vertical with feet on the floor) over a 24 hour period provides insight to their disease severity and degree of functional impairment. Our goal was to turn this self-report questionnaire about upright activity into a wearable device that could accurately and passively measure upright activity. We coined this measure “UpTime”.
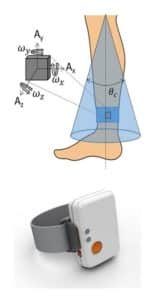
This research was funded by the Bateman Horne Center, the University of Utah, and with a Ramsey Award from Solve ME/CFS to Shad Roundy.

 Lucinda Bateman, MD, is a renowned clinician, researcher, and educator. Her Johns Hopkins University Medical School training instilled an approach to care that she has employed throughout her career - the patient comes first and the unknown or unexplained does not equate to a lack of proper and compassionate care. Since starting her own practice in 2000, she has served on six boards or committees, been the principal investigator for 45 studies, authored/coauthored 40 journal articles, served as adjunct instructor and adjunct assistant professor in the University of Utah Departments of Preventative Medicine, Internal Medicine, and Anesthesiology, and lectured around the world.
Lucinda Bateman, MD, is a renowned clinician, researcher, and educator. Her Johns Hopkins University Medical School training instilled an approach to care that she has employed throughout her career - the patient comes first and the unknown or unexplained does not equate to a lack of proper and compassionate care. Since starting her own practice in 2000, she has served on six boards or committees, been the principal investigator for 45 studies, authored/coauthored 40 journal articles, served as adjunct instructor and adjunct assistant professor in the University of Utah Departments of Preventative Medicine, Internal Medicine, and Anesthesiology, and lectured around the world.